Home » What causes rusting when metal products encounter corrugated boxes?

A Corrugated box maker was asked by his customer to supply boxes made from acid and chloride free, low Cobb kraft paper as they would be packing steel automotive parts. The box maker’s problem was, how to convert this specification of kraft paper
from qualitative parameters to quantitative parameters.
A packaged food manufacturer received complaints from market that some of the tin containers showed rusting on the rim of metal container which was in contact with corrugated box.

All views and insights expressed in this blog are contributed by Mr. Ramkumar Sunkara.
We come across such issues quite often. Some of the typical corrosion related issues are :
1. Corrosion of tin containers when packed in corrugated box.
2. Corrosion of metal parts when packed directly into box.
3. Tarnishing of polished metal parts when wrapped in paper.
This article highlights the various quality parameters of kraft paper that need to be specified and their significance from point of view of preventing corrosion reaction
when a metal product is in contact with it. The parameters responsible for initiating rusting or tarnishing of metal parts, when they are in contact with kraft paper are :
1. Moisture levels in the paper.
2. pH of Paper.
3. Porosity and Oil Absorbency of paper.
4. Chloride levels in paper.
5. Sulphate levels in paper.
6. Microbiological influence of the paper.
The mechanism of initiation of corrosive reaction due to each of the above parameters is discussed below.
1. Moisture levels in the paper.
Corrosion is an electrochemical process, requiring the presence of an electrolyte. Moisture in the paper provides the essential electrolyte required for electrochemical
corrosion reaction. When a certain critical humidity level is reached, the moisture in the paper tends to form a thin film (invisible) electrolyte on the surface of the metal. For example, in case of iron this critical humidity level is around 60%. This moisture in form of thin film is extremely critical from the point of view of the chemical mechanism of the corrosion process.
The “time of wetness” (TOW) of a corroding surface is a key parameter, directly determining the duration for the start of the electrochemical corrosion process. The
TOW refers to the period during which the conditions are favourable for the formation of a surface layer of moisture on a metal or alloy. The TOW is strongly dependent on the critical relative humidity levels. Kraft paper being hygroscopic material, it absorbs moisture from air and loses moisture to the air. The rate of absorption is dependent on the amount of moisture in the air (Rh) and its temperature while the rate of loss is dependent on the moisture content of the paper and its temperature.
Paper holds moisture either between the fibres or within the individual fibres as shown in the figure.
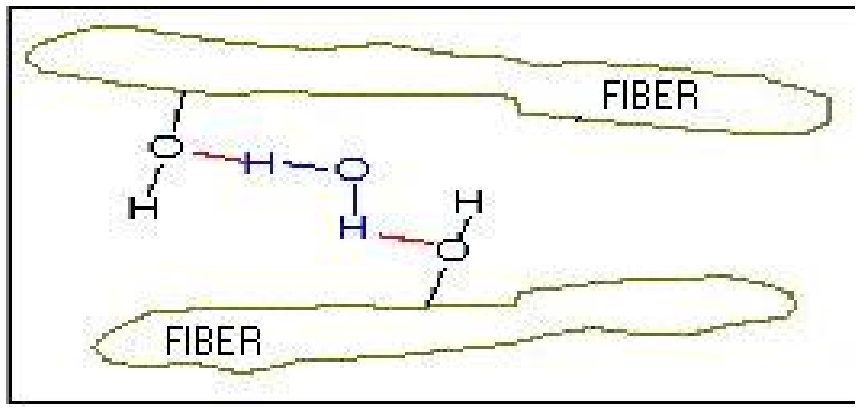
H2O molecule forms the bond between the individual fibers. Moisture between the fibres can be subdivided into :
a. Colloidal water.
b. Capillary water
At low vapour pressure (Low Rh) most of the water is present as colloidal water. (Water absorbed at the free hydroxyls) At high moisture content (High Rh)
considerable capillary water (Water absorbed in the pores and voids) is present. It is this capillary water, which tends form the surface layer (thin film) of moisture on the metal surface when it is in contact with paper.
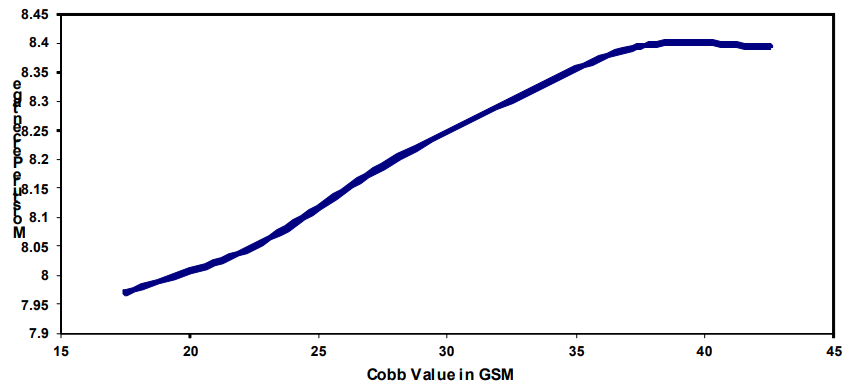
At the same Rh, paper with low Cobb value has less capillary water as compared to paper with high Cobb value. A graph of Cobb Vs moisture percentage of paper at 65% Rh and 27oC is shown below.
For example,
| Cobb of paper (65% RH & 27°C) |
Moisture percentage at | ||
|---|---|---|---|
| 30% RH | 65% RH | 90% RH | |
| 20 | 5.7 | 7.9 | 11.6 |
| 45 | 5.6 | 8.6 | 13.2 |
This The shows higher moisture percentage at the same Rh for paper with higher Cobb value above 60% Rh. Use of paper with higher Cobb value will lead to faster film
formation of moisture at Rh above 60% on the surface of the metal.
Hence care must be taken to ensure that Kraft paper used for metal wrapping is having Cobb value less than 25.
Corrugated boards are a sandwich structure of liners and medium. The main job of the medium is to maintain the parallelism of the two liners. Media must maintain this separation of liners to form the sandwich. (Fig 1)
2. pH (Acidity) of paper :
Low pH accelerates corrosion by providing plentiful supply of hydrogen ions. Paper usually has pH in the range from 5 to 8. In this range of pH, paper is non-corrosive.
The threshold level of Hydrogen ion content (pH) in unbleached kraft paper, which is in contact with steel sheets, is 5. Kraft papers used for wrapping of metal parts having its pH lower than 5 will lead to serious corrosion risk. One must ensure that pH of paper is between 6.5 and 7.5 (Acid free paper), if the paper is encountering metal parts.
3.Porosity and Oil absorbency of paper :
Porosity of paper is related to the oil resistance of paper. It is observed that the most oil-resistant papers have very low porosity. There is linear relation between air resistance measured in seconds on the Gurley instrument and oil resistance in seconds. The relationship is :
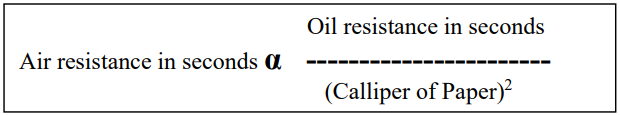
Wrapping paper should have very low porosity to prevent penetration of Hydrogen Sulphide, Oxygen, Carbon dioxide, and other gases that cause tarnishing of steel.
Low porosity will lead to low oil absorbency. This will help in maintaining oil film on the metal parts intact preventing rusting or tarnishing of metal parts.
4. Chloride levels in paper :
Chloride ion significantly accelerates corrosion. Chloride ions also cause localized break down of the passive film (Anti-rust oil film) on the metal part. Chloride ions increase the electrical conductivity of the water so that the flow of corrosion currents is facilitated. Chloride ions reduce the effectiveness of natural protective films. Chloride ions are responsible for the initiation and propagation of localized corrosion damage. Soluble salts like chlorides and Sulphates pull moisture from air and cause failure of protective coating. Chlorides are found naturally in paper because of water recycling, use of secondary fibre, pulping chemicals etc. The threshold limit for chloride content in unbleached non-corrosive kraft paper is 0.05%. Kraft paper used for this application must have chloride level as NaCl % below 0.05%
5. Sulphate levels in paper :
As compared to the corrosive effect of chloride ion levels, Sulphates are generally considered to be more benign in their corrosive action towards metallic materials. Sulphate ions formed in the surface moisture layer are responsible for accelerating corrosion rather than the corrosion itself. The presence of sulphates some time does pose a major risk for metallic materials in the sense that sulphates can be converted to highly corrosive sulphides by anaerobic sulphate reducing bacteria. The threshold limit for Sulphate content in unbleached non-corrosive kraft paper is 0.25%. Kraft paper used for this application must have sulphate level as Na2SO4 % below 0.25%.
6. Microbiological influence of paper :
Bacteria, Fungi, and other micro-organisms can play major part in corrosion of metals. Microbiologically Influenced Corrosion (MIC) refers to corrosion that is influenced by the presence and activities of micro-organisms and/or their metabolites (the products produced in their metabolism processes). Microbiological surface colonies on the paper lead to the formation of corrosion pits, driven by microbial activity and associated with the location of these colonies.
Micro-organism associated with corrosion damage are classified as :
a. Anaerobic bacteria (bacteria which do not require or cannot tolerate oxygen in their life process) that produce highly corrosive by-products as part of their metabolism.
b. Aerobic bacteria (bacteria requiring oxygen for life process) that produce corrosive mineral acids.
c. Fungi that may produce corrosive by-products in their metabolism, such as organic acids. Bacteria tend to live and grow under wide range of temperature, pH and oxygen concentration in paper. Carbon molecule (from paper making starches) is the source of nutrient for bacteria. Sulphate reducing bacteria (SRB) are anaerobes that are sustained by organic nutrients present in paper. Most common strains of SRB grow best at temperatures from 250C to 350C. SRB reduces Sulphate to Sulphide, which usually shows up as hydrogen sulphide or if iron is available, then as black ferrous sulphide. The key symptom that usually indicates SRB involvement in the corrosion process of ferrous alloys is localized corrosion filled with black sulphide corrosion products. Anti-fungal and anti-bacterial agents must be added to pulp at the time of paper making to prevent growth of micro-organisms.